 *Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu #Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No LrĬlick on the element to view its propertiesĮlement (sym) # Mass density oxid #'s mp bp EN Ionization Energy rad. Where #M# is the molar mass in grams per mole ( #gmol^-1#), #m# is the mass of the sample in grams ( #g#), and #n# is the amount in moles of the sample ( #mol#).The Periodic Table of the Elements H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La* Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac#. 
Ask your teacher or tutor if you are not sure which one to use.Īnother way to find molar mass if amount and mass of a substance are known is by using the formula The molar masses will be slightly different if you are using a table with more accurate decimal values for atomic mass. Sulfuric acid #H_"2"SO_4# has 2 Hydrogen atoms (mass #1#), 1 sulfur atom (mass #32#) and 4 oxygen atoms (mass #16#) so: Then simply find out how many of each atom is in the molecule and add the respective atomic masses to find the molecular or molar mass.įor example water #H_2O# has 2 Hydrogen atoms (mass #1#) and 1 oxygen atom (mass #16#) so: 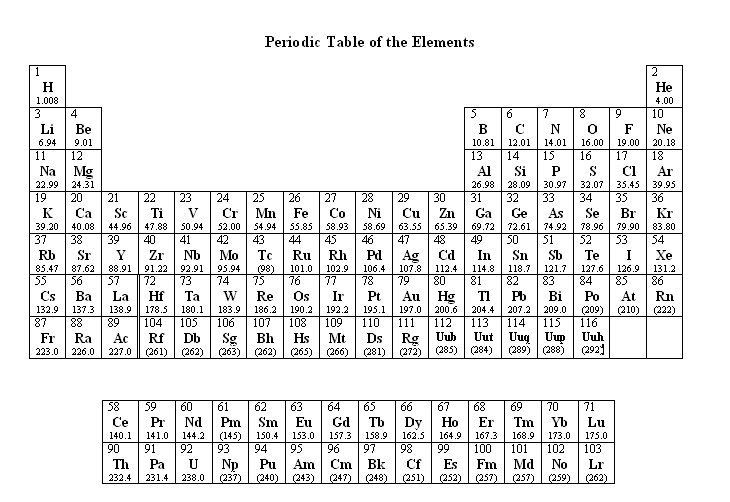
I prefer to use tables with whole number atomic masses, but that is just personal preference. Find a periodic table and look at the atomic mass of each atom in the molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
